
Scan the QR
to read the magazine on mobile phone
Big shots gather to discuss major infectious disease prevention and control challenges
On the afternoon of July 13th, "Major Infectious Disease Prevention and Control Forum" was held in Yantai. The forum is organized by Luye Life Sciences, and was an important part of the "International Conference on Pharmaceutical Innovation and Development 2023".
Five heavyweight guests from disease control, immunization and drug delivery were invited to share the latest research results in their respective fields, explore the challenges of major infectious disease prevention and control, and look forward to industry development trends.
During the round-table session on "Insight into Biomedical Products", guests expressed their opinions on topics such as how to make innovation in vaccines to meet the challenges of public health and clinical needs; and how to strengthen cooperation between different departments and organizations to improve overall response capabilities.
Mindfront Health Awarded the "Demonstration Organization" by the National Mental Health Specialist Alliance
From July 14th to 16th, 2023, the first Forum on Psychiatry for Universal Conversation and Innovation was held in Shanghai. During the forum, 12 "Demonstration Unit" of the National Medical Center for Psychiatric Diseases and the Specialty Alliance of Shanghai Mental Health Centers were announced. Mindfront was the only private specialised medical institution in the winner’s list.

The Puci Forum on Mental Health is hosted by the National Center for Psychiatry, Shanghai Mental Health Center, and Shanghai Mental Health Society. Besides Mindfront Health, 11 public mental health institutions Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai First People's Hospital, Shanghai Children's Hospital, Shanghai Hongkou District Mental Health Center, and public medical institutions from Xinjiang, Yunnan, Shenzhen and other places accepted the award.
Ms. Liu Aona, Executive Chairperson of Luye Medical Group and Founding Director of Mindfront Health said: "Being rated as a 'demonstration Unit' by the National Mental Health Specialty Alliance is not only a compliment, but also a responsibility and mission. Mindfront Health will always be people-oriented, give full play to its own characteristics and advantages, and contribute to the ‘Healthy China’ blueprint by supporting the construction of the "dual centers" of the National Medical Center and the National Regional Medical Center."

Yantai Huayi Rehabilitation Hospital Attended the Yantai 2023 International Conference on Pharmaceutical Innovation and Development
At the Yantai 2023 International Conference on Pharmaceutical Innovation and Development held on 13th July, Iris Shi, Vice President of Operation of Luye Medical Group and Head of Yantai Campus was invited to deliver a keynote speech at the High-end Medical Devices Branch, talking about a new path for rehabilitation services and facilities in China.

She started with the development trend of rehabilitation medicine and made a systematic exposition on how the hospital localized world-class rehabilitation services and met demands for rehabilitation medical equipment. Through the analysis of the status quo of the rehabilitation medical industry and the reference to the development of the rehabilitation industry in the Us, she pointed out that there is currently a huge gap in domestic rehabilitation medical beds, talents, and rehabilitation equipment, and the development potential is strong. Then, through research on the classification and market size of rehabilitation medical equipment, she shared the future demand trend of the rehabilitation equipment industry and looked forward to the development opportunities of rehabilitation equipment based on different rehabilitation stages and scenarios.
Iris said Huayi Rehabilitation Hospital will spare no efforts on improving the quality of rehabilitation services and optimizing high-end equipment, escorting the health of more people with professional medical technology, and making new contributions to the development of Yantai's rehabilitation and high-end medical equipment industry.

Luye Diagnostics Releases China’s First Non-Invasive Diagnostic Kits for H. pylori and Its DRMs
Luye Diagnostics hosted a product launch event on 28th July centered around the theme "Preventing Gastric Hazards and Living a H. pylori-Free Life" at the Shanghai International Convention Center. Concurrently, a Symposium on Diagnostics for Digestive Diseases was also held. During this event, the company introduced its independently developed non-invasive diagnostic kits for detecting H. pylori and its drug-resistant mutations (DRMs). These groundbreaking products include an H. pylori 23S rRNA gene mutation detection kit (PCR-fluorescence probe method), which stands as China's first non-invasive H. pylori DRM testing product utilizing fecal samples, alongside an H. pylori detection kit (PCR-fluorescence probe method).
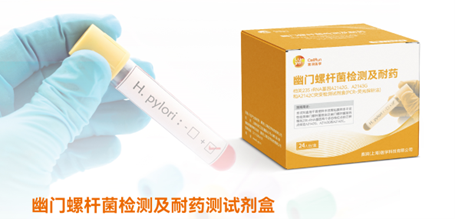
H. pylori 23S rRNA gene mutation detection kit (PCR-fluorescence probe method)
Luye Diagnostics' H. pylori 23S rRNA gene mutation detection kit (PCR-fluorescence probe method) marks China's pioneering effort in developing a domestically produced kit for the non-invasive detection of H. pylori DRMs using fecal samples. This cutting-edge kit effectively addresses the current challenges of H. pylori resistance testing. Patients who are H. pylori positive or suspected of such infection can conveniently collect fecal samples at home and send them back for testing. Unlike the traditional culture-based H. pylori drug sensitivity testing, which takes 7 to 10 days, this product completes the testing within merely 2 hours. Offering the advantages of user-friendly operation, swift turnaround time, and high sensitivity, it proves ideal for initial clinical DRM screening. Armed with the drug resistance testing results, doctors can administer targeted treatment to enhance the success rate of H. pylori eradication.

H. pylori detection kit (PCR-fluorescence probe method)
The H. pylori detection kit (PCR-fluorescence probe method) offers a swift and non-invasive solution for H. pylori detection, rendering it highly suitable for home-based testing and general screening. This product enables the detection of H. pylori infection in both fecal and gastric mucosal samples, allowing for convenient self-sampling at home. As a non-invasive test with minimal risk, it proves user-friendly and appropriate for diverse patient groups, including those planning to conceive, pregnant women, and children.

Ms. Ella Jiang, the CEO of Luye Diagnostics, noted, “Cancer prevention and control are priorities of the Healthy China 2030 Plan. Experts specializing in early gastric cancer screening unanimously recognize H. pylori as a relevant biomarker for such screenings. H. pylori need not be feared; rather, early diagnosis, timely treatment, and appropriate medication are the keys to managing it effectively. The newly released products combine the advantages of non-invasive sampling and DRM testing, alleviating patient discomfort during testing and enabling doctors to prescribe targeted treatments based on drug resistance results, thus offering a precision medicine-based solution to H. pylori-related gastric problems. These rapid and non-invasive detection kits are ideal for home-based testing and general screening, helping prevent the transmission of H. pylori within families and avoiding scenarios where one person's infection leads to the infection of the entire family. As a result, they hold significant clinical value and play a crucial role in safeguarding public health. We hope that these products would benefit numerous patients with digestive tract diseases and contribute to reducing the incidence of gastric cancer in the country.”

